"This unprecedented meningitis outbreak in Kent underscores the devastating speed of bacterial infection and prompts a critical re-evaluation of vaccine policy for teenagers, highlighting the complex interplay of public health, epidemiology, and resource allocation."
The recent cluster of bacterial meningitis cases in Kent, which has tragically claimed two lives and affected dozens, has sent shockwaves across the UK. Labelled "unprecedented" by health authorities due to its rapid spread and severity, this outbreak serves as a stark reminder of the devastating potential of meningococcal disease. While proactive measures, including mass antibiotic distribution, are underway to contain its spread, the event has triggered urgent questions about national vaccine strategies, the bacterium’s evolving nature, and potential environmental or social factors contributing to such a swift escalation.
Bacterial meningitis, a severe infection of the membranes surrounding the brain and spinal cord, is a medical emergency that can progress with terrifying speed. One day, an individual can be in peak health; the next, they may be fighting for their life in intensive care as bacteria invade vital neurological systems and poison the bloodstream, a condition known as septicaemia. While invasive bacterial meningitis has become rare in the UK thanks to widespread vaccination programmes, the emergence of a cluster of this magnitude in Kent has presented an acute public health crisis.
The first case linked to this outbreak was reported to the UK Health Security Agency (UKHSA) on 13 March, leading to public warnings issued just two days later. What followed was a rapid escalation, with 29 people now confirmed or suspected to have been affected, resulting in two fatalities. This scale and speed of transmission, particularly within a short timeframe and localised geography, is what health officials have termed "unprecedented," prompting a comprehensive, multi-faceted response. As the situation continues to evolve, several critical questions remain at the forefront of the public health discourse.
When Will This Be Over?
The immediate concern for residents and public health officials alike is the containment and eventual cessation of the outbreak. The reporting of only two new cases on Friday, while cautiously encouraging, does not signify the end of the crisis. Given the typical incubation period for meningococcal disease, which can range from two to ten days, further cases are anticipated as individuals who were exposed earlier manifest symptoms.
Epidemiological investigations have identified a "super-spreader event" at Club Chemistry in Canterbury between 5 March and 7 March as the likely ignition point for the cluster. Super-spreader events, where an unusually large number of individuals are infected by a single source or during a specific gathering, are notoriously difficult to control due to the high density of contacts. The ten-day incubation window means that, hopefully, the majority of cases directly linked to those specific nights in the nightclub have already emerged or are in the process of doing so.
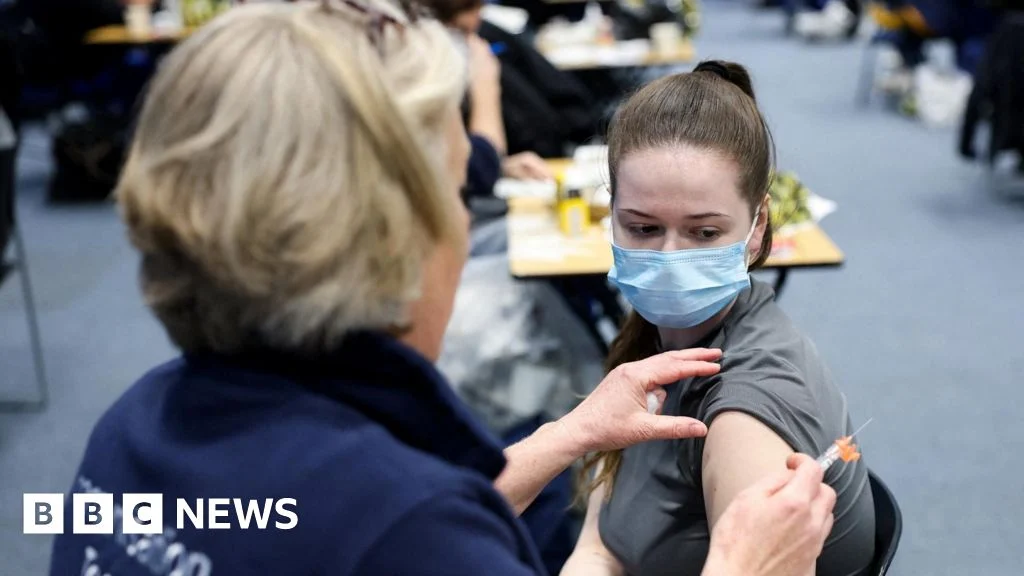
A cornerstone of the public health response has been the rapid distribution of antibiotics to approximately 10,000 individuals identified as close contacts. This prophylactic measure is crucial, as these antibiotics are designed to eliminate the meningitis bacteria from carriers, preventing them from developing the disease themselves or transmitting it further. While these efforts are extensive and proactive, their full impact will only become evident over time, as the incubation period concludes for the broader exposed population and surveillance continues to track new infections. Public health officials face a delicate balance between managing public anxiety and providing transparent, evidence-based updates until the outbreak is definitively declared over.
Will It Spread Beyond Kent?
Currently, all confirmed and suspected cases have a direct epidemiological link to Kent, particularly the Canterbury area. However, the interconnectedness of modern society, especially within student populations, raises concerns about potential wider geographical spread. Reports indicate that some students left their university residences to return home in other parts of the country when news of the outbreak broke.
The Neisseria meningitidis bacteria, responsible for meningococcal disease, can reside harmlessly in the nose and throat of up to 25% of adolescents and young adults without causing illness. These asymptomatic carriers can, however, transmit the bacteria to others through close contact, such as prolonged face-to-face interaction, kissing, or sharing utensils. Therefore, there is a distinct possibility that an asymptomatic carrier may have travelled outside of Kent, potentially passing on the bacteria to new contacts in other regions.
A monumental contact tracing effort is underway, with over 10,000 potential close contacts identified and offered prophylactic antibiotics. This scale of tracing is unprecedented for a bacterial meningitis outbreak in the UK. Should new cases emerge outside of Kent, it will be critical for public health authorities to distinguish between those genetically linked to the current outbreak strain and the approximately one case of invasive bacterial meningitis that occurs daily across the UK, which would be unrelated. Vigilant surveillance and rapid genomic sequencing will be key to understanding the true geographical reach of this specific strain.
Should All Teenagers Be Given the Vaccine?
The outbreak has inevitably reignited the debate surrounding the MenB vaccine and its inclusion in the routine vaccination schedule for adolescents. Health Secretary Wes Streeting has formally requested that the government’s vaccine advisors, the Joint Committee on Vaccination and Immunisation (JCVI), re-evaluate the evidence.

The efficacy of the MenB vaccine is not in question; it is highly effective at preventing disease caused by meningococcus serogroup B. The core of the debate, however, revolves around cost-effectiveness and the optimal allocation of NHS resources. The MenB vaccine is considerably expensive, costing approximately £220 for the full course if sought privately.
When the JCVI first assessed the MenB vaccine over a decade ago, it concluded that it was cost-effective for infants and toddlers, the age group at highest risk of severe disease, and was subsequently introduced into the routine childhood immunisation programme. However, at that time, the committee determined it was not cost-effective for universal vaccination of teenagers and young adults, who are also at elevated risk but experience a lower incidence of disease compared to infants.
The current review will critically examine whether the epidemiological landscape has shifted sufficiently to alter these mathematical calculations. The "unprecedented" nature of this outbreak, with its rapid spread and severe outcomes within a young adult population, could potentially tip the balance. The JCVI’s decision will involve a complex assessment of the direct health benefits of preventing disease, the indirect benefits of reducing transmission (herd immunity), and the economic impact on the healthcare system, all weighed against the societal burden of the disease. This is a cold mathematical calculation, often feeling distant from the immense pain experienced by families devastated by meningitis, yet it is fundamental to national public health policy.
Has the Bacterium Become More Dangerous?
A crucial aspect of understanding and controlling the outbreak lies in characterizing the pathogen itself. An initial genetic analysis of the bacterium responsible for the Kent outbreak, completed on Thursday, revealed that it is similar to strains that have been circulating in the UK since 2021. Critically, this initial assessment suggests that the existing MenB vaccine should offer protection against this particular strain.
However, "similar" does not mean identical, and more detailed genetic analysis is now underway. Even small mutations within a bacterial genome can significantly alter its phenotype – its observable characteristics. These changes could impact the bacterium’s transmissibility, making it easier to spread from person to person; its invasiveness, increasing its ability to penetrate host tissues and cause systemic disease; or its virulence, leading to more severe illness. Experts will be scrutinizing the whole genome sequence to identify any such mutations that might explain the rapid and severe nature of this outbreak. Understanding these genetic nuances is vital for refining public health interventions and informing future vaccine strategies.
What Else Could Have Triggered Such a Rapid Outbreak?
Beyond the intrinsic properties of the bacterium, epidemiologists are exploring external factors that might have contributed to the unprecedented scale and speed of this outbreak. Several hypotheses are being considered:
-
Vape Sharing and Close Contact Behaviors: The social environment of a packed nightclub, often involving close physical proximity and shared items like vapes, could facilitate the rapid transmission of respiratory pathogens. While sharing vapes is unhygienic, and has been a topic of public "chatter," there is no definitive scientific proof directly linking it to this outbreak’s scale, though it aligns with the known routes of meningococcal transmission via respiratory droplets.
-
"Immunity Gap" from Covid Lockdowns: A more compelling hypothesis relates to the potential impact of Covid-19 lockdowns. Prolonged periods of reduced social mixing could have led to a cohort of teenagers and young adults with less natural exposure to common pathogens, including Neisseria meningitidis. Typically, many individuals are exposed to and carry meningococcal bacteria asymptomatically, building a level of natural immunity. Reduced exposure during lockdowns might have created an "immunity gap," leaving this age group more susceptible to infection when social interactions resumed. This phenomenon has been observed with other respiratory viruses, such as RSV and influenza, post-lockdown.
-
Saharan Dust Cloud: An intriguing environmental factor that has been pointed out is the timing of a Saharan dust cloud sweeping over Europe and the UK, coinciding with the initial period of the outbreak. Dust particles, particularly fine particulate matter, are known to irritate the respiratory airways, potentially compromising the delicate mucous membranes and local immune defenses. This irritation could make it easier for meningococcal bacteria, normally residing harmlessly in the nasopharynx, to invade the bloodstream and cause invasive disease. This mechanism is well-established in the "Meningitis Belt" of sub-Saharan Africa, where seasonal dust storms are a known risk factor for meningitis outbreaks.
It is entirely plausible that there is no single, definitive cause, but rather a confluence of multiple smaller factors that, when combined, created the perfect storm for such a rapid and severe outbreak. This complexity underscores the challenges inherent in epidemiological investigation and the multi-factorial nature of infectious disease dynamics.
Ongoing Vigilance and Future Implications
As the UKHSA and local health authorities continue their intensive efforts to contain the Kent meningitis outbreak, public vigilance remains paramount. Recognizing the symptoms of meningitis and septicaemia – including a sudden high fever, headache, stiff neck, dislike of bright lights, drowsiness, joint pain, and a distinctive rash that does not fade under pressure – is crucial for early diagnosis and treatment, which are vital for improving outcomes.
This unprecedented event will undoubtedly have long-term implications for public health policy. The re-evaluation of the MenB vaccine for adolescents by the JCVI could lead to significant changes in the national immunization schedule. Furthermore, the outbreak serves as a potent reminder of the fragility of public health security and the continuous need for robust surveillance, rapid response mechanisms, and adaptability in the face of evolving infectious disease threats. The lessons learned from Kent will be critical in shaping future preparedness strategies against rare but devastating illnesses.