This groundbreaking UK research, successfully demonstrating fully functional, lab-grown oesophagi in animal models, heralds a new era of hope for children born with severe oesophageal defects, promising a future of fewer surgeries and a dramatically improved quality of life.
The ability to bioengineer complex organs has long been a medical aspiration, particularly for conditions where natural tissue is critically absent or severely damaged. For infants like two-year-old Casey Mcintyre, born with a significant portion of his oesophagus missing, life has been a relentless series of interventions and challenges. Now, scientists in the UK have achieved a remarkable feat, cultivating fully functioning food pipes in the laboratory and successfully transplanting them into mini pigs, a breakthrough published in the prestigious journal Nature Biotechnology that could revolutionize treatment for this devastating congenital condition.
Oesophageal atresia (OA) is a rare congenital anomaly where a baby’s oesophagus, the tube connecting the mouth to the stomach, does not develop properly, resulting in a gap. In severe cases, known as long-gap oesophageal atresia, the segment missing can be substantial, making surgical connection extremely difficult or impossible. Approximately 18 babies are born in the UK each year with a similar severe condition to Casey, facing a lifetime of complex medical care. Current treatments often involve multiple, invasive surgeries to attempt to bridge the gap, such as gastric pull-up (moving the stomach higher into the chest) or colon interposition (using a segment of the colon to create a new oesophagus). These procedures carry significant risks, including strictures, chronic reflux, and long-term feeding difficulties, often necessitating feeding tubes for years and impacting a child’s overall development and quality of life.
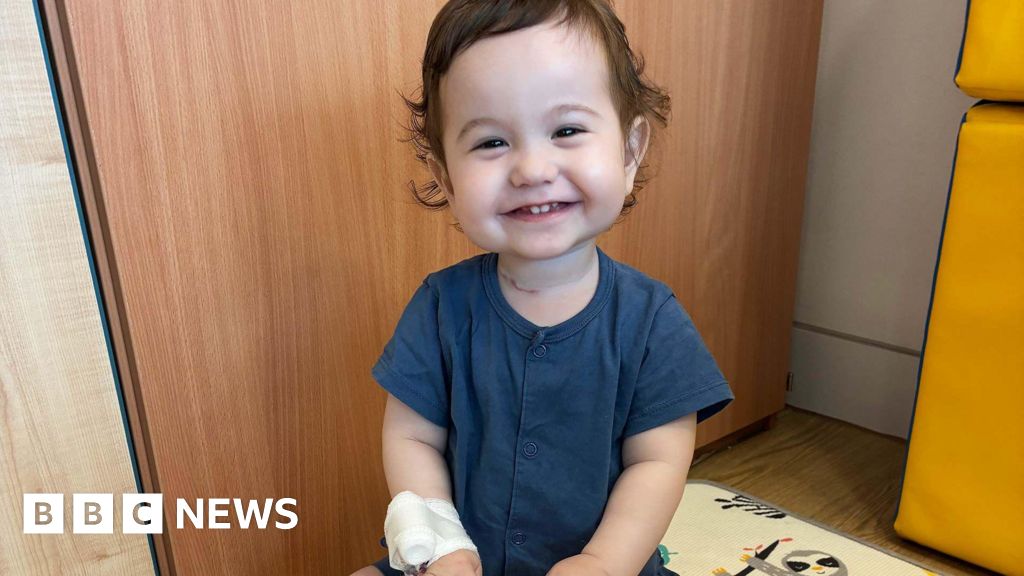
Casey Mcintyre’s journey exemplifies the profound challenges faced by families grappling with this condition. Born with an 11cm segment of his oesophagus missing, his parents, Silviya and Sean, were informed of his major issues before birth, bracing themselves for extensive medical interventions. Doctors performed a complex surgery to reposition his stomach to bridge the significant gap. Yet, the road to recovery has been arduous. Casey continues to rely on a feeding tube, and repeated surgeries have unfortunately resulted in damage to his vocal cords, delaying his speech development. His mother, Silviya, explains the family’s anticipation: "Once he’s eating enough through his mouth, we’ll be able to take his tube out." Sean adds that they have had to acquire skills far beyond typical new parent experiences, from managing a stomach tube to navigating urgent hospital calls. Despite these challenges, they remain immensely proud of Casey, recognizing the "miracle" of his current care, but envisioning a future where a single, early operation could replace the missing oesophagus, providing a definitive solution that would be "life-changing."
The scientific team, led by Professor Paolo De Coppi at Great Ormond Street Hospital (GOSH) and University College London (UCL), has addressed these challenges with an innovative tissue engineering approach. Their method involves a two-stage process: decellularization and recellularization, followed by maturation in a bioreactor. First, a donor oesophagus from a pig is subjected to a sophisticated decellularization process, which carefully strips away all cellular material, including any immunogenic components, while preserving the intricate three-dimensional extracellular matrix (ECM) – the natural scaffold of the organ. This remaining biological framework provides the essential structural and biochemical cues necessary for new tissue growth, without triggering an immune rejection response.
Next, this decellularized scaffold is repopulated with the recipient animal’s own cells, a process known as recellularization. While the original article specifies "the animals own cells," this typically involves harvesting progenitor cells or stem cells from the patient themselves, ensuring perfect immunological compatibility. These cells are seeded onto the scaffold, which is then placed in a specialized bioreactor. This bioreactor is a meticulously controlled environment that mimics the physiological conditions of the body, continuously pumping vital growth fluids, nutrients, and providing mechanical stimulation (such as pulsatile flow and gentle stretching) to encourage cell proliferation, differentiation, and tissue maturation over approximately one week. This dynamic environment is crucial for guiding the seeded cells to develop into the complex cell types found in a functional oesophagus, including epithelial lining, smooth muscle layers for peristalsis, and nerve cells for proper control.
The research deliberately utilized Göttingen minipigs, a breed specifically chosen for their remarkable physiological and anatomical similarities to human children, particularly in terms of size and cellular makeup. This makes them an ideal translational model for pediatric applications. Eight minipigs underwent transplantation of these lab-grown oesophagi. The results were highly encouraging: all eight pigs recovered well, demonstrating the critical development of working swallowing muscles. Crucially, the implants integrated seamlessly into the animals’ bodies, with five pigs surviving to the six-month endpoint of the trial, where their grafts exhibited fully functional muscle, nerves, and blood vessels. A significant advantage of using the animals’ own cells for recellularization was the complete absence of any need for anti-rejection drugs, a major concern with traditional organ transplantation.
Professor De Coppi emphasized the inherent complexity of the oesophagus, noting it is "a really complex organ, without a blood supply from its own vessels, so it cannot be ‘transplanted’ in the way you might expect." This inherent challenge underscores the ingenuity of the tissue engineering approach, which effectively creates a living, self-sustaining organ. He articulated his ambitious but hopeful vision of offering this treatment to children within the next five years. This timeline reflects the need for further rigorous pre-clinical validation, navigating stringent regulatory approval processes from bodies like the Medicines and Healthcare products Regulatory Agency (MHRA), and developing robust surgical protocols for human application. A key benefit for pediatric patients is that the bioengineered oesophagus is designed to grow with the child, eliminating the need for subsequent surgical enlargements or replacements that would be necessary with static implants.

However, De Coppi also clarified the current limitations of this specific graft. It is not currently suitable for adults with other oesophageal problems, such as those arising from cancer. This is primarily due to the size mismatch – the current grafts are scaled for children – and the differing regenerative and reconstructive needs of adult pathologies, which often involve extensive tissue removal and different vascularization challenges.
This pioneering work stands as a testament to the transformative potential of regenerative medicine. It pushes the boundaries of tissue engineering, moving closer to a future where damaged or missing organs can be grown from a patient’s own cells, eliminating the need for organ donors and the lifelong immunosuppression associated with allogeneic transplants. While the immediate focus is on long-gap oesophageal atresia, the underlying principles and techniques developed in this research could potentially be applied to bioengineer other hollow organs, offering hope for countless patients facing similar debilitating conditions. The journey from lab to clinic is often long and complex, but for families like the McIntyres, this scientific breakthrough shines a bright light on a future where children born with severe oesophageal defects can look forward to a healthier, more normal life, free from the burden of continuous medical interventions.
